| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Ethene[1] | |||
| Systematic IUPAC name
Ethene | |||
| Other names
Refrigerant R-1150
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| 1730731 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.000.742 | ||
| EC Number |
| ||
| 214 | |||
| KEGG | |||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1962 1038 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C 2H 4 | |||
| Molar mass | 28.054 g·mol−1 | ||
| Appearance | colourless gas | ||
| Density | 1.178 kg/m3 at 15 °C, gas[2] | ||
| Melting point | −169.2 °C (−272.6 °F; 104.0 K) | ||
| Boiling point | −103.7 °C (−154.7 °F; 169.5 K) | ||
| 131 mg/L (25 °C);[3] 2.9 mg/L[4] | |||
| Solubility in ethanol | 4.22 mg/L[4] | ||
| Solubility in diethyl ether | good[4] | ||
| Acidity (pKa) | 44 | ||
| Conjugate acid | Ethenium | ||
| −15.30·10−6 cm3/mol | |||
| Viscosity | 10.28 μPa·s[5] | ||
| Structure | |||
| D2h | |||
| zero | |||
| Thermochemistry | |||
Std molar
entropy (S⦵298) |
219.32 J·K−1·mol−1 | ||
Std enthalpy of
formation (ΔfH⦵298) |
+52.47 kJ/mol | ||
| Hazards | |||
| GHS labelling: | |||
 
| |||
| Danger | |||
| H220, H336 | |||
| P210, P261, P271, P304+P340, P312, P377, P381, P403, P403+P233, P405, P501 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | −136 °C (−213 °F; 137 K) | ||
| 542.8 °C (1,009.0 °F; 815.9 K) | |||
| Safety data sheet (SDS) | ICSC 0475 | ||
| Related compounds | |||
Related compounds
|
Ethane Acetylene Propene | ||
| Supplementary data page | |||
| Ethylene (data page) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
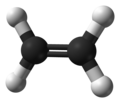
Ethylene (IUPAC name: ethene) is a hydrocarbon which has the formula C2H4 or H2C=CH2. It is a colourless, flammable gas with a faint "sweet and musky" odour when pure.[7] It is the simplest alkene (a hydrocarbon with carbon–carbon double bonds).
Ethylene is widely used in the chemical industry, and its worldwide production (over 225 million tonnes in 2022)[8] exceeds that of any other organic compound.[9][10] Much of this production goes toward creating polyethylene, which is a widely used plastic containing polymer chains of ethylene units in various chain lengths. Production emits greenhouse gases, including methane from feedstock production and carbon dioxide from any non-sustainable energy used.
Ethylene is also an important natural plant hormone and is used in agriculture to induce ripening of fruits.[11] The hydrate of ethylene is ethanol.
Structure and properties

This hydrocarbon has four hydrogen atoms bound to a pair of carbon atoms that are connected by a double bond. All six atoms that comprise ethylene are coplanar. The H-C-H angle is 117.4°, close to the 120° for ideal sp² hybridized carbon. The molecule is also relatively weak: rotation about the C-C bond is a very low energy process that requires breaking the π-bond by supplying heat at 50 °C.[citation needed]
The π-bond in the ethylene molecule is responsible for its useful reactivity. The double bond is a region of high electron density, thus it is susceptible to attack by electrophiles. Many reactions of ethylene are catalyzed by transition metals, which bind temporarily to the ethylene using both the π and π* orbitals.[citation needed]
Being a simple molecule, ethylene is spectroscopically simple. Its UV-vis spectrum is still used as a test of theoretical methods.[12]
Uses

Major industrial reactions of ethylene include in order of scale: 1) polymerization, 2) oxidation, 3) halogenation and hydrohalogenation, 4) alkylation, 5) hydration, 6) oligomerization, and 7) hydroformylation. In the United States and Europe, approximately 90% of ethylene is used to produce ethylene oxide, ethylene dichloride, ethylbenzene and polyethylene.[13] Most of the reactions with ethylene are electrophilic addition.[citation needed]

Polymerization
Polyethylene production uses more than half of the world's ethylene supply. Polyethylene, also called polyethene and polythene, is the world's most widely used plastic. It is primarily used to make films in packaging, carrier bags and trash liners. Linear alpha-olefins, produced by oligomerization (formation of short-chain molecules) are used as precursors, detergents, plasticisers, synthetic lubricants, additives, and also as co-monomers in the production of polyethylenes.[13]
Oxidation
Ethylene is oxidized to produce ethylene oxide, a key raw material in the production of surfactants and detergents by ethoxylation. Ethylene oxide is also hydrolyzed to produce ethylene glycol, widely used as an automotive antifreeze as well as higher molecular weight glycols, glycol ethers, and polyethylene terephthalate.[14]
Ethylene oxidation in the presence of a palladium catalyst can form acetaldehyde. This conversion remains a major industrial process (10M kg/y).[15] The process proceeds via the initial complexation of ethylene to a Pd(II) center.[citation needed]
Halogenation and hydrohalogenation
Major intermediates from the halogenation and hydrohalogenation of ethylene include ethylene dichloride, ethyl chloride, and ethylene dibromide. The addition of chlorine entails "oxychlorination", i.e. chlorine itself is not used. Some products derived from this group are polyvinyl chloride, trichloroethylene, perchloroethylene, methyl chloroform, polyvinylidene chloride and copolymers, and ethyl bromide.[16]
Alkylation
Major chemical intermediates from the alkylation with ethylene is ethylbenzene, precursor to styrene. Styrene is used principally in polystyrene for packaging and insulation, as well as in styrene-butadiene rubber for tires and footwear. On a smaller scale, ethyltoluene, ethylanilines, 1,4-hexadiene, and aluminium alkyls. Products of these intermediates include polystyrene, unsaturated polyesters and ethylene-propylene terpolymers.[16]
Oxo reaction
The hydroformylation (oxo reaction) of ethylene results in propionaldehyde, a precursor to propionic acid and n-propyl alcohol.[16]
Hydration
Ethylene has long represented the major non-fermentative precursor to ethanol. The original method entailed its conversion to diethyl sulfate, followed by hydrolysis. The main method practiced since the mid-1990s is the direct hydration of ethylene catalyzed by solid acid catalysts:[17]
- C2H4 + H2O → CH3CH2OH
Dimerization to butenes
Ethylene is dimerized by hydrovinylation to give n-butenes using processes licensed by Lummus or IFP. The Lummus process produces mixed n-butenes (primarily 2-butenes) while the IFP process produces 1-butene. 1-Butene is used as a comonomer in the production of certain kinds of polyethylene.[18]
Fruit and flowering
Ethylene is a hormone that affects the ripening and flowering of many plants. It is widely used to control freshness in horticulture and fruits.[19] The scrubbing of naturally occurring ethylene delays ripening.[20] Adsorption of ethylene by nets coated in titanium dioxide gel has also been shown to be effective.[21]
Niche uses
An example of a niche use is as an anesthetic agent (in an 85% ethylene/15% oxygen ratio).[22] It is also used as a refrigerant gas for low temperature applications under the name R-1150.[23]
Production
Global ethylene production was 107 million tonnes in 2005,[9] 109 million tonnes in 2006,[24] 138 million tonnes in 2010, and 141 million tonnes in 2011.[25] By 2013, ethylene was produced by at least 117 companies in 32 countries. To meet the ever-increasing demand for ethylene, sharp increases in production facilities are added globally, particularly in the Mideast and in China.[26] Production emits greenhouse gas, namely significant amounts of carbon dioxide.[27]
Industrial process
Ethylene is produced by several methods in the petrochemical industry. A primary method is steam cracking (SC) where hydrocarbons and steam are heated to 750–950 °C. This process converts large hydrocarbons into smaller ones and introduces unsaturation. When ethane is the feedstock, ethylene is the product. Ethylene is separated from the resulting mixture by repeated compression and distillation.[16] In Europe and Asia, ethylene is obtained mainly from cracking naphtha, gasoil and condensates with the coproduction of propylene, C4 olefins and aromatics (pyrolysis gasoline).[28] Other procedures employed for the production of ethylene include Fischer-Tropsch synthesis and methanol-to-olefins (MTO).[29]
Laboratory synthesis
Although of great value industrially, ethylene is rarely synthesized in the laboratory and is ordinarily purchased.[30] It can be produced via dehydration of ethanol with sulfuric acid or in the gas phase with aluminium oxide or activated alumina.[31]
Biosynthesis
Ethylene is produced from methionine in nature. The immediate precursor is 1-aminocyclopropane-1-carboxylic acid.[32]
Ligand

Ethylene is a fundamental ligand in transition metal alkene complexes. One of the first organometallic compounds, Zeise's salt is a complex of ethylene. Useful reagents containing ethylene include Pt(PPh3)2(C2H4) and Rh2Cl2(C2H4)4. The Rh-catalysed hydroformylation of ethylene is conducted on an industrial scale to provide propionaldehyde.[34]
History
Some geologists and scholars believe that the famous Greek Oracle of Delphi (the Pythia) went into her trance-like state as an effect of ethylene rising from ground faults.[35]
Ethylene appears to have been discovered by Johann Joachim Becher, who obtained it by heating ethanol with sulfuric acid;[36] he mentioned the gas in his Physica Subterranea (1669).[37] Joseph Priestley also mentions the gas in his Experiments and observations relating to the various branches of natural philosophy: with a continuation of the observations on air (1779), where he reports that Jan Ingenhousz saw ethylene synthesized in the same way by a Mr. Enée in Amsterdam in 1777 and that Ingenhousz subsequently produced the gas himself.[38] The properties of ethylene were studied in 1795 by four Dutch chemists, Johann Rudolph Deimann, Adrien Paets van Troostwyck, Anthoni Lauwerenburgh and Nicolas Bondt, who found that it differed from hydrogen gas and that it contained both carbon and hydrogen.[39] This group also discovered that ethylene could be combined with chlorine to produce the Dutch oil, 1,2-dichloroethane; this discovery gave ethylene the name used for it at that time, olefiant gas (oil-making gas.)[40] The term olefiant gas is in turn the etymological origin of the modern word "olefin", the class of hydrocarbons in which ethylene is the first member.[citation needed]
In the mid-19th century, the suffix -ene (an Ancient Greek root added to the end of female names meaning "daughter of") was widely used to refer to a molecule or part thereof that contained one fewer hydrogen atoms than the molecule being modified. Thus, ethylene (C
2H
4) was the "daughter of ethyl" (C
2H
5).[citation needed]. The name ethylene was used in this sense as early as 1852.[41]
In 1866, the German chemist August Wilhelm von Hofmann proposed a system of hydrocarbon nomenclature in which the suffixes -ane, -ene, -ine, -one, and -une were used to denote the hydrocarbons with 0, 2, 4, 6, and 8 fewer hydrogens than their parent alkane.[42] In this system, ethylene became ethene. Hofmann's system eventually became the basis for the Geneva nomenclature approved by the International Congress of Chemists in 1892, which remains at the core of the IUPAC nomenclature. However, by that time, the name ethylene was deeply entrenched, and it remains in wide use today, especially in the chemical industry.
Following experimentation by Luckhardt, Crocker, and Carter at the University of Chicago,[43] ethylene was used as an anesthetic.[44][7] It remained in use through the 1940s, even while chloroform was being phased out. Its pungent odor and its explosive nature limit its use today.[45]
Nomenclature
The 1979 IUPAC nomenclature rules made an exception for retaining the non-systematic name ethylene;[46] however, this decision was reversed in the 1993 rules,[47] and it remains unchanged in the newest 2013 recommendations,[48] so the IUPAC name is now ethene. In the IUPAC system, the name ethylene is reserved for the divalent group -CH2CH2-. Hence, names like ethylene oxide and ethylene dibromide are permitted, but the use of the name ethylene for the two-carbon alkene is not. Nevertheless, use of the name ethylene for H2C=CH2 (and propylene for H2C=CHCH3) is still prevalent among chemists in North America.[49]
Safety
Like all hydrocarbons, ethylene is a combustible asphyxiant. It is listed as an IARC group 3 agent, since there is no current evidence that it causes cancer in humans.[50]
See also
- RediRipe, an ethylene detector for fruits.
References
- ^ "Ethylene". Archived from the original on 2023-10-08. Retrieved 2021-05-27.
- ^ Record of Ethylene in the GESTIS Substance Database of the Institute for Occupational Safety and Health, accessed on 25 October 2007.
- ^ McAuliffe, C. (1966). "Solubility in Water of Paraffin, Cycloparaffin, Olefin, Acetylene, Cycloolefin, and Aromatic Hydrocarbons". Journal of Physical Chemistry. 70 (4): 1267–1275. Bibcode:1966JPhCh..70.1267M. doi:10.1021/j100876a049.
- ^ a b c Neiland, O. Ya. (1990) Органическая химия: Учебник для хим. спец. вузов. Moscow. Vysshaya Shkola. p. 128.
- ^ Kestin J, Khalifa HE, Wakeham WA (1977). "The viscosity of five gaseous hydrocarbons". The Journal of Chemical Physics. 66 (3): 1132–1134. Bibcode:1977JChPh..66.1132K. doi:10.1063/1.434048.
- ^ ETHYLENE | CAMEO Chemicals | NOAA Archived 2015-04-02 at the Wayback Machine. Cameochemicals.noaa.gov. Retrieved on 2016-04-24.
- ^ a b Zimmermann H, Walz R (2008). "Ethylene". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a10_045.pub3. ISBN 978-3527306732.
- ^ "Decarbonization approaches for ethylene production: comparative techno-economic and life-cycle analysis". Royal Society of Chemistry. Royal Society of Chemistry. Retrieved 13 November 2025.
- ^ a b "Production: Growth is the Norm". Chemical and Engineering News. 84 (28): 59–236. July 10, 2006. doi:10.1021/cen-v084n034.p059.
- ^ Propylene Production from Methanol. Intratec. 2012-05-31. ISBN 978-0-615-64811-8. Archived from the original on 2016-03-04. Retrieved 2012-09-17.
- ^ Wang KL, Li H, Ecker JR (2002). "Ethylene biosynthesis and signaling networks". The Plant Cell. 14 (Suppl): S131-151. Bibcode:2002PlanC..14S.131W. doi:10.1105/tpc.001768. PMC 151252. PMID 12045274.
- ^ "Ethylene:UV/Visible Spectrum". NIST Webbook. Archived from the original on 2017-01-19. Retrieved 2006-09-27.
- ^ a b "OECD SIDS Initial Assessment Profile — Ethylene" (PDF). inchem.org. Archived from the original (PDF) on 2015-09-24. Retrieved 2008-05-21.
- ^ "Ethylene Glycol: Systemic Agent". Center for Disease Control. 20 October 2021. Archived from the original on 26 December 2017. Retrieved 20 February 2022.
- ^ Elschenbroich C, Salzer A (2006). Organometallics: A Concise Introduction (2nd ed.). Weinheim: Wiley-VCH. ISBN 978-3-527-28165-7.
- ^ a b c d Kniel L, Winter O, Stork K (1980). Ethylene, keystone to the petrochemical industry. New York: M. Dekker. ISBN 978-0-8247-6914-7.
- ^ Kosaric N, Duvnjak Z, Farkas A, Sahm H, Bringer-Meyer S, Goebel O, Mayer D (2011). "Ethanol". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. pp. 1–72. doi:10.1002/14356007.a09_587.pub2. ISBN 9783527306732.
- ^ "1-Butene - Major Uses". WISER. Archived from the original on Nov 16, 2021. Retrieved 2021-11-16.
- ^ Arshad, Muhammad; Frankenberger, William (2002). Ethylene. Boston, MA: Springer. p. 289. ISBN 978-0-306-46666-3.
- ^ Melton, Laurence; Shahidi, Fereidoon; Varelis, Peter (2019). Encyclopedia of Food Chemistry. Netherlands: Elsevier. p. 114. ISBN 978-0-12-814045-1.
- ^ de Matos Fonseca, J.; Pabón, N.Y.L; Nandi, L.G (2021). "Gelatin-TiO2-coated expanded polyethylene foam nets as ethylene scavengers for fruit postharvest application". Postharvest Biology and Technology. 180 111602. doi:10.1016/j.postharvbio.2021.111602.
- ^ Trout HH (August 1927). "Blood Changes Under Ethylene Anæsthesia". Annals of Surgery. 86 (2): 260–7. doi:10.1097/00000658-192708000-00013. PMC 1399426. PMID 17865725.
- ^ "R-1150 ETHYLENE Safety Data Sheet" (PDF). Australian Refrigeration Mechanics Association. April 2015. Archived (PDF) from the original on 1 July 2023. Retrieved 1 July 2023.
- ^ Nattrass, L and Higson, A (22 July 2010) NNFCC Renewable Chemicals Factsheet: Ethanol Deprecated link archived 2012-09-05 at archive.today. National Non-Food Crops Centre
- ^ True WR (2012). "Global ethylene capacity poised for major expansion". Oil & Gas Journal. 110 (7): 90–95. Archived from the original on 2016-06-04. Retrieved 2016-05-06.
- ^ "Market Study: Ethylene (2nd edition), Ceresana, November 2014". ceresana.com. Archived from the original on 2015-03-07. Retrieved 2015-02-03.
- ^ Mynko, Oleksii; Amghizar, Ismaël; Brown, David J.; Chen, Lin; Marin, Guy B.; de Alvarenga, Rodrigo Freitas; Uslu, Didem Civancik; Dewulf, Jo; Van Geem, Kevin M. (2022-08-15). "Reducing CO2 emissions of existing ethylene plants: Evaluation of different revamp strategies to reduce global CO2 emission by 100 million tonnes". Journal of Cleaner Production. 362 132127. Bibcode:2022JCPro.36232127M. doi:10.1016/j.jclepro.2022.132127. hdl:1854/LU-8760240. ISSN 0959-6526. S2CID 248838079.
- ^ "Ethylene Production and Manufacturing Process". Icis. Archived from the original on 2019-07-29. Retrieved 2019-07-29.
- ^ Amghizar I, Vandewalle LA, Van Geem KM, Marin GB (2017). "New Trends in Olefin Production". Engineering. 3 (2): 171–178. Bibcode:2017Engin...3..171A. doi:10.1016/J.ENG.2017.02.006.
- ^ Crimmins MT, Kim-Meade AS (2001). "Ethylene". In Paquette, L. (ed.). Encyclopedia of Reagents for Organic Synthesis. New York: Wiley. doi:10.1002/047084289X.re066. ISBN 0471936235.
- ^ Cohen JB (1930). Practical Organic Chemistry (preparation 4). Macmillan.
- ^ Yang SF, Hoffman NE (1984). "Ethylene biosynthesis and its regulation in higher plants". Annu. Rev. Plant Physiol. 35: 155–89. doi:10.1146/annurev.pp.35.060184.001103.
- ^ Neely, Jamie M. (2014). "chlorobis(ethylene)rhodium(I) dimer". E-EROS Encyclopedia of Reagents for Organic Synthesis. pp. 1–6. doi:10.1002/047084289X.rn01715. ISBN 9780470842898.
- ^ Wiley-VCH, ed. (2003-03-11). Ullmann's Encyclopedia of Industrial Chemistry (1 ed.). Wiley. doi:10.1002/14356007.a22_157.pub3. ISBN 978-3-527-30385-4. Archived from the original on 2018-03-05. Retrieved 2023-10-17.
- ^ Roach J (2001-08-14). "Delphic Oracle's Lips May Have Been Loosened by Gas Vapors". National Geographic. Archived from the original on September 24, 2001. Retrieved March 8, 2007.
- ^ Roscoe HE, Schorlemmer C (1878). A treatise on chemistry. Vol. 1. D. Appleton. p. 611.
- ^ Brown JC (July 2006). A History of Chemistry: From the Earliest Times Till the Present Day. Kessinger. p. 225. ISBN 978-1-4286-3831-0.
- ^ Appendix, §VIII, pp. 474 ff., Experiments and observations relating to the various branches of natural philosophy: with a continuation of the observations on air, Joseph Priestley, London: printed for J. Johnson, 1779, vol. 1.
- ^ Roscoe & Schorlemmer 1878, p. 612
- ^ Roscoe & Schorlemmer 1878, p. 613
Gregory W (1857). Handbook of organic chemistry (4th American ed.). A.S. Barnes & Co. p. 157. - ^ "ethylene | Etymology, origin and meaning of ethylene". etymonline. Archived from the original on 2022-07-19. Retrieved 2022-07-19.
- ^ Hofmann AW. "Hofmann's Proposal for Systematic Nomenclature of the Hydrocarbons". www.chem.yale.edu. Archived from the original on 2006-09-03. Retrieved 2007-01-06.
- ^ Luckhardt A, Carter JB (1 December 1923). "Ethylene as a gas anesthetic". Current Researches in Anesthesia & Analgesia. 2 (6): 221–229. doi:10.1213/00000539-192312000-00004. S2CID 71058633.
- ^ Johnstone GA (August 1927). "Advantages of Ethylene-Oxygen as a General Anesthetic". California and Western Medicine. 27 (2): 216–8. PMC 1655579. PMID 18740435.
- ^ Whalen FX, Bacon DR, Smith HM (September 2005). "Inhaled anesthetics: an historical overview". Best Practice & Research. Clinical Anaesthesiology. 19 (3): 323–30. doi:10.1016/j.bpa.2005.02.001. PMID 16013684.
- ^ IUPAC nomenclature rule A-3.1 (1979) Archived 2000-10-10 at the Wayback Machine. Acdlabs.com. Retrieved on 2016-04-24.
- ^ Footnote to IUPAC nomenclature rule R-9.1, table 19(b) Archived 2007-12-19 at the Wayback Machine. Acdlabs.com. Retrieved on 2016-04-24.
- ^ Favre, Henri A.; Powell, Warren H., eds. (2014). Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013. Cambridge: Royal Society of Chemistry. ISBN 9781849733069. OCLC 865143943.
- ^ Vollhardt, K. Peter C.; Schore, Neil Eric (2018). Organic chemistry : structure and function (8 ed.). New York: Macmillan Learning. p. 470. ISBN 978-1-319-07945-1. OCLC 1007924903.
- ^ "Ethylene (IARC Summary & Evaluation, Volume 60, 1994)". www.inchem.org. Archived from the original on 2019-01-13. Retrieved 2019-01-13.